Corneal Cross-linking
This is the newest treatment for keratoconus that has reduced the number of people that will need surgery for the condition. Keratoconus is a progressive disease that results in thinning and weakening of the cornea. Corneal Cross-Linking is a procedure that employs the use of Vitamin B2 (Riboflavin) and a special light to strengthen the cornea to try and prevent thinning. It is important to note that the purpose of Corneal Cross-Linking is not to improve the vision, but instead to stop the progression of the disease. Your doctor will determine if you are a good candidate for this procedure.
How it works:
- Creates new corneal collagen cross-links
- Results in a shortening and thickening on the collagen fibrils
- Leads to the stiffening of the cornea
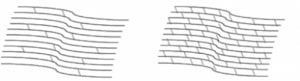
Less Cross-Linking (Weaker) More Cross-Linking (Stronger)
What to Expect:
This FDA approved treatment takes approximately 70 minutes to complete in office. You do not need any anesthesia for this procedure, and it is not painful, although you may experience some mild discomfort and light sensitivity in the days following the procedure while your eye is healing.
Riboflavin
Riboflavin (vitamin B2) functions as a photo-enhancer which enables the cross- linking reaction to occur. It is a drop administered to the eye every minute for 60 minutes.
Ultra-Violet A Light (UVA)
UVA is one of the three types of invisible light rays given off by the sun (together with ultra-violet B and ultra-violet C) and is the weakest of the three. A UV light source is applied to irradiate the cornea after it has been soaked in the photo enhancing riboflavin solution. This cross-linking process stiffens the cornea by increasing the number of molecular bonds, or cross-links, in the collagen.